Latest News
-
May 31, 2026Ufafanuzi wa Changamoto za Mfumo Mpya wa Pamoja wa Ugomboaji wa Mizigo (TANESW)
-
May 21, 2026Ban to Manufacture, Import, Supply or Sale Veterinary Pharmaceutical Product Namely - Hi-tet 120
-
May 21, 2026Existence of Unregistered Human Medicinal Product with Brand Name VEGA – 100 Tablets
-
Apr 20, 2026Disposal of Expired and Unfit Medical Products
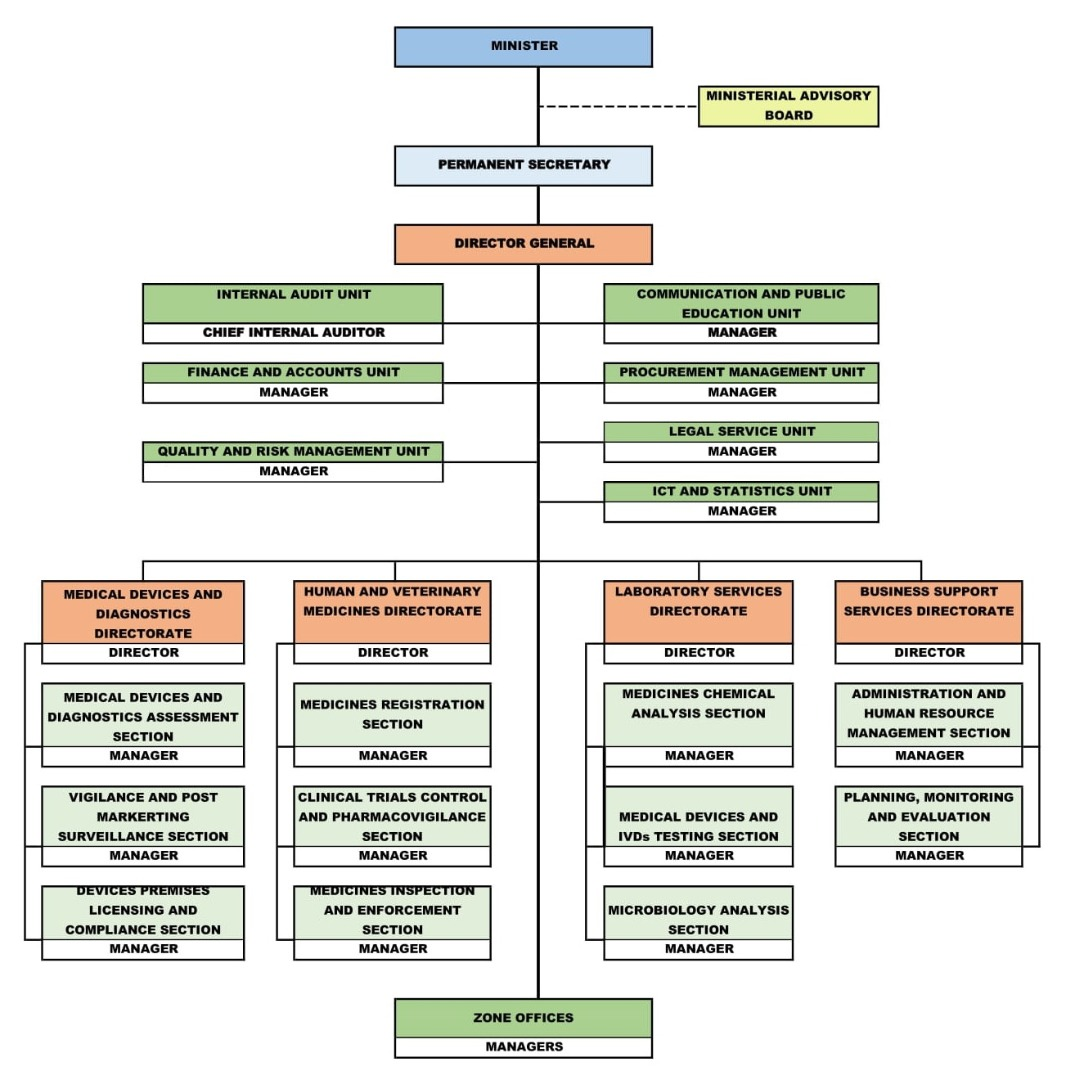
TMDA Organogram
Approved by the President of the United Republic of Tanzania on 2021